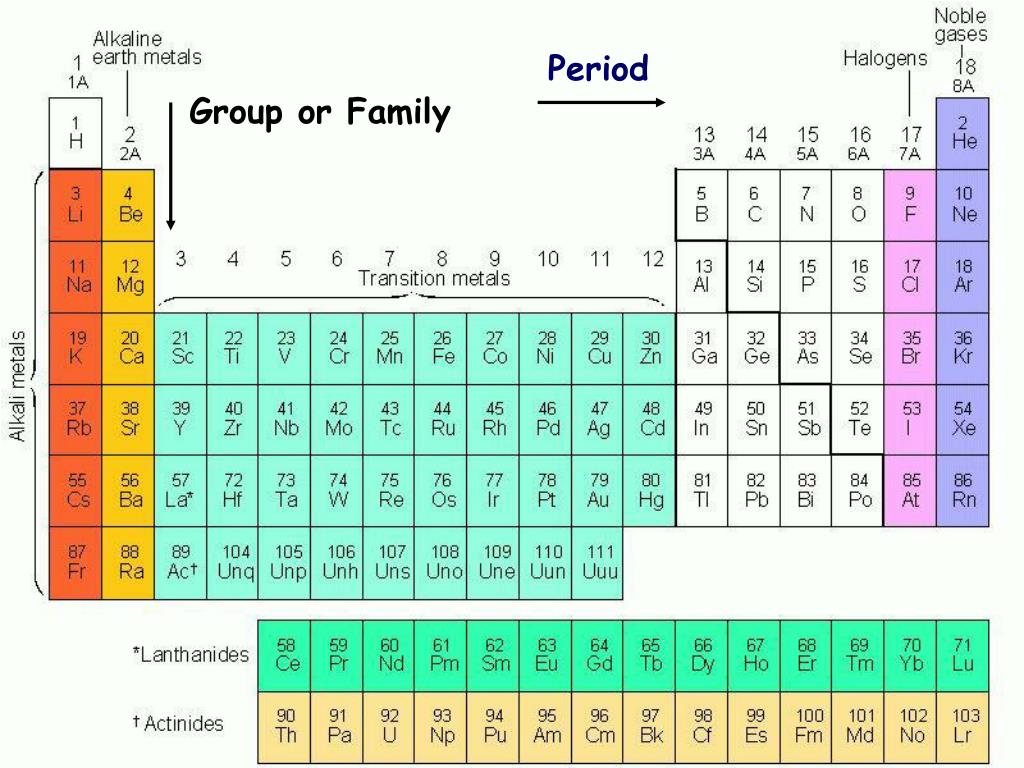
Transition Metals are the most number of elements in the periodic table. They consist of Beryllium (Be), Magnesium (Mg), Calcium (Ca), Strontium (Sr), Barium (Ba), Radium (Ra) The most reactive metals consisting of Lithium (Li), Sodium (Na), Potassium (K), Rubidium (Rb), Cesium (Cs), and Francium (Fr)Īlkaline Earth Metals Group 18: the noble gases ( helium/ neon family).Group 17: the halogens ( fluorine family).Group 16: the chalcogens ( oxygen family).Group 15: the pnictogens ( nitrogen family).Group 2: the alkaline earth metals ( beryllium family).Group 1: the alkali metals ( lithium family) *not including hydrogen.The other ten groups are transition metals: There are eight main groups of elements, numbered 1, 2, and 13-18. Elements in the same group usually have similar properties, because they have the same number of electrons in the outermost electron shell. ( January 2022)Ī group is any column on the periodic table. You can help Wikipedia by reading Wikipedia:How to write Simple English pages, then simplifying the article. The English used in this article or section may not be easy for everybody to understand. For the story of the periodic table, you need to go to the next quiz. A Russian scientist named Dmitri Mendeleev placed the elements in order of atomic mass whereas we now use atomic number, which was unknown at the time. Many scientists made attempts at this, including a teacher called John Dalton and a French chemist called Alexandre-Emile Beguyer de Chancourtois (what a name!!). In the 1800s, chemists were regularly finding new elements and they desperately needed a way to organise them to see the patterns that were becoming evident. Unfortunately, Lavoisier was a nobleman and as a result, during the French Revolution, he was murdered by the revolutionaries - he was guillotined in Paris. His contribution to the periodic table was to classify his list of elements as metals and non metals. His list of elements contained substances such as oxygen, nitrogen, mercury and zinc but there were also some compounds that he believed to be elements, plus light and heat, which at the time were still believed to be materials rather than forms of energy. 
In it, he began the search to classify the elements in a meaningful way. Twenty years after this, French scientist Antoine de Lavoisier wrote what is regarded as the first chemistry textbook. In the middle of the seventeenth century, Irish scientist Robert Boyle came up with a definition of an element that lasted until the the discovery of sub-atomic particles, he said that "an element is a substance that could not be broken down into a simpler substance by a chemical reaction". 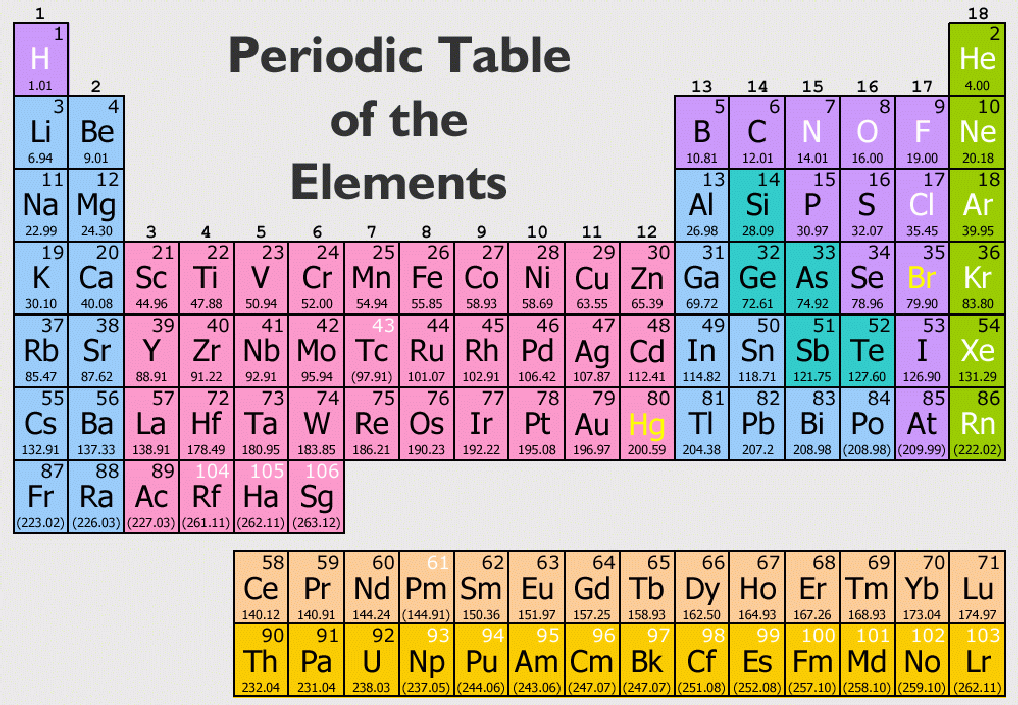
It would have been a very difficult task anyway because some of the substances they discovered were compounds only a few of them were what we now know are true elements. They managed to discover some of the materials that made up the world around them, however, since they were actually looking for a mythical substance that would turn base metals into gold (the Philosopher's Stone), they never actually thought of devising a periodic table. It's a long way from our current model of over 100 elements arranged into 8 groups but there are some people who stiill believe this mystical idea! It took 1,000 years before the alchemists of Islam started to carry out experiments systematically. 
They believed that everything was made from four elements - fire, air, earth and water. The ancient Greek philosophers knew very little about science, however, that didn't stop them coming up with ideas to help to explain the world around them. In this quiz we take a look at how the periodic table is organised into groups and periods, and also at some of the information we can get about elements from their position in the table. For GCSE Chemistry it is vital that students have a good understanding of the periodic table.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
